
However, when Erwin Schrödinger sent a preprint of his new equation to Einstein, Einstein wrote back hailing his equation as a decisive advance of “true genius.” But the next shock came in 1926 when Max Born proposed that mechanics were to be understood as a probability without any causal explanation.īoth Einstein and Erwin Schrödinger rejected this interpretation with its renunciation of causality which had been a key feature of science previous to Quantum Mechanics and was still a feature of General Relativity. The shocks for Einstein began in 1925 when Werner Heisenberg introduced matrix equations that removed the Newtonian elements of space and time from any underlying reality. The quantum revolution of the mid-1920s occurred under the direction of both Einstein and Bohr, and their post-revolutionary debates were about making sense of the change. However, Einstein was right and Bohr proved to be wrong about light quanta. Bohr fought back against the existence of the quantum of light (photon) by writing the BKS theory in 1924. Einstein had proposed the photon in 1905, and Compton proved that the photon existed experimentally in 1922, but Bohr refused to believe the photon existed even then. This was perhaps the first real Bohr-Einstein debate. Bohr did not like a scientist having to choose between equations. 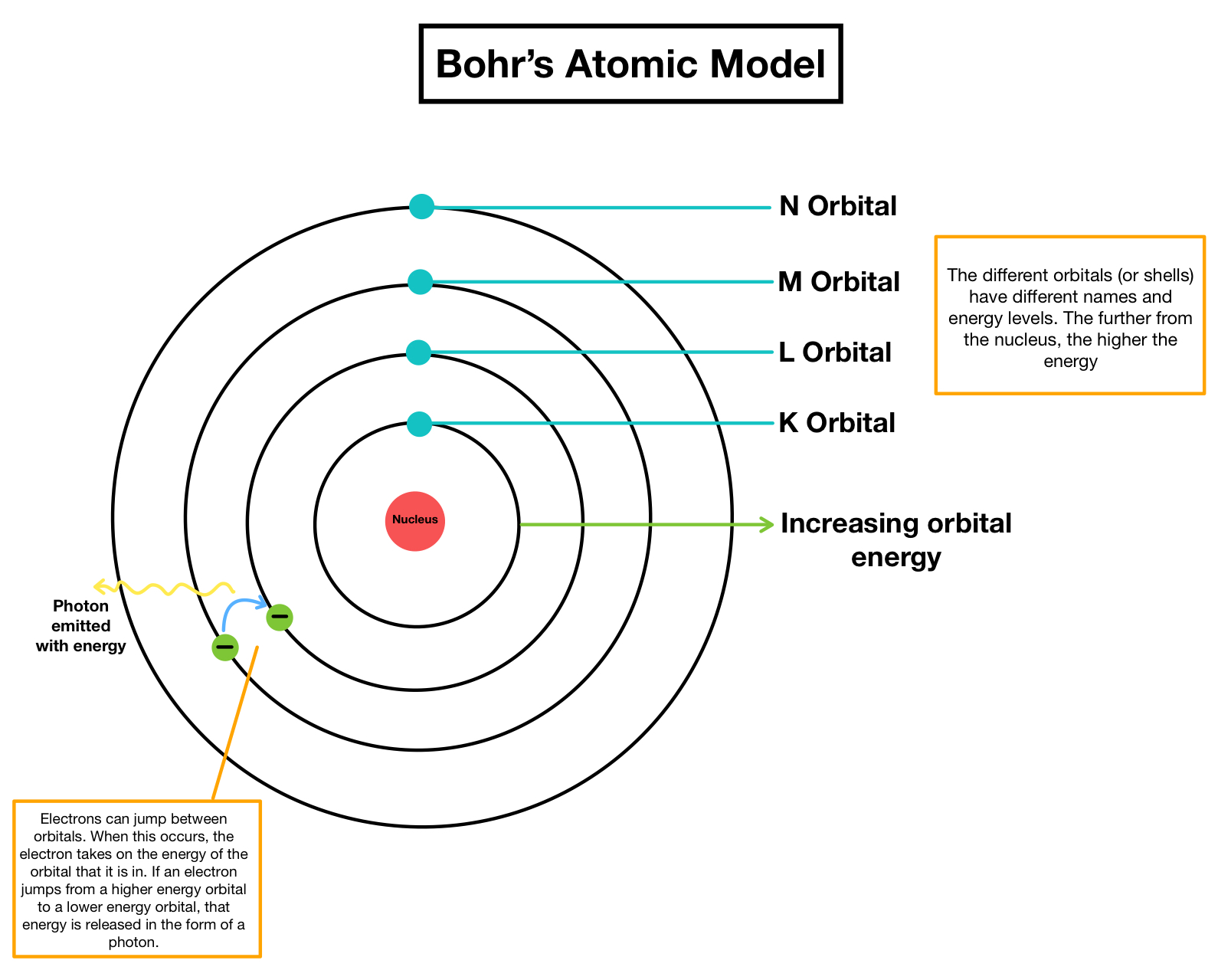
Bohr disliked it because it made the choice of mathematical solution arbitrary. The photon appealed to Einstein because he saw it as a physical reality (although a confusing one) behind the numbers presented by Planck mathematically in 1900. 
Bohr was one of the most vocal opponents of the photon idea and did not openly embrace it until 1925. To support his point, in 1905 he proposed that light sometimes acts as a particle which he called a light quantum (see photon and wave–particle duality). Pre-revolutionary debates Įinstein was the first physicist to say that Planck's discovery of the quantum ( h) would require a rewriting of the laws of physics. Īlthough Bohr and Einstein disagreed, they were great friends all their lives and enjoyed using each other as a foil. Despite their differences of opinion and the succeeding discoveries that helped solidify quantum mechanics, Bohr and Einstein maintained a mutual admiration that was to last the rest of their lives. Based on the article, the philosophical issue of the debate was whether Bohr's Copenhagen Interpretation of quantum mechanics, which centered on his belief of complementarity, was valid in explaining nature. On Epistemological Problems in Atomic Physics". Most of Bohr's version of the events held in Solvay in 1927 and other places was first written by Bohr decades later in an article titled, "Discussions with Einstein Their debates are remembered because of their importance to the philosophy of science, insofar as the disagreements-and the outcome of Bohr's version of quantum mechanics becoming the prevalent view-form the root of the modern understanding of physics. The Bohr–Einstein debates were a series of public disputes about quantum mechanics between Albert Einstein and Niels Bohr. That is a reason not to teach about circular orbits and L = nħ.Series of public disputes between physicists Niels Bohr and Albert Einstein Niels Bohr (left) with Albert Einstein (right) at Paul Ehrenfest's home in Leiden (December 1925) But second, it doesn’t conform to the essence of what Bohr actually did. This, in itself, is not a reason to scrap it, for the historical development of quantum physics is certainly of interest. First it doesn’t conform to our present understanding of the hydrogen atom. This picture of the hydrogen atom is wrong in two senses. This “pedagogic” version postulates that an electron in a stationary state moves in a circular orbit with an angular momentum that is an integral multiple of h/2π ( L = nh/2π = nħ)- ħ for the lowest-energy state, 2 ħ for the next state, and so on. Unfortunately, the version of the Bohr atom that appears in many textbooks and is no doubt often presented to students is more wrong than right and may leave both teachers and students wondering why, more than a hundred years later, it is still being taught. This is an excellent idea, given the historic importance of Bohr’s 1913 work, which provided the bridge from Planck’s quantized interaction of matter and radiation (1900) to the full theory of quantum mechanics (1925-28). Many teachers like to introduce the Bohr atom toward the end of an introductory physics course.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
